Home >> Uncategorized >> How to write a protocol for a clinical trial
How to write a protocol for a clinical trial
Apr/Mon/2017 | Uncategorized
WHO | Recommended format for a Research Protocol

Writing a Protocol | CHOP Institutional Review Board

Protocol guidance and template for use in a Clinical Trial of an

Key elements to writing a strong clinical trial - Protocol Builder
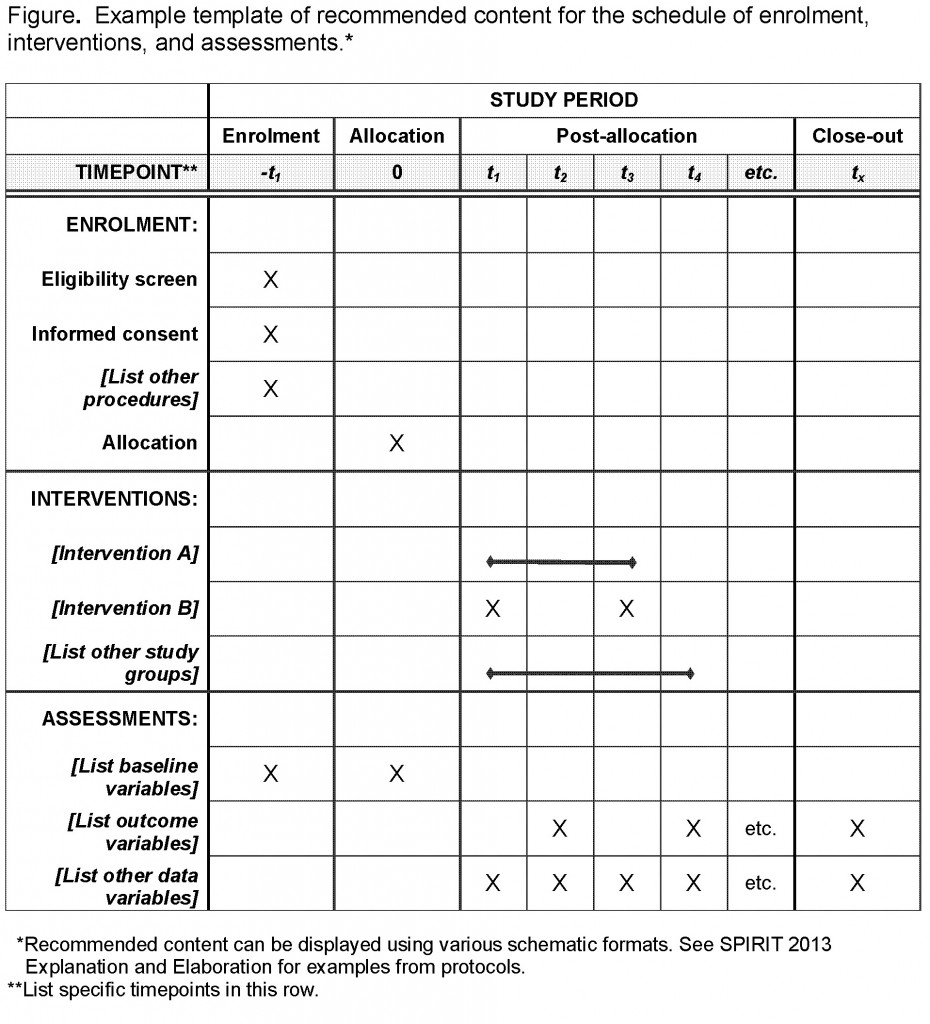
Effectively Writing Clinical Trial Protocols - Barnett Educational Services

How to design and write a clinical research protocol in Cosmetic

FDA and NIH Release a Draft Clinical Trial Protocol Template for

Key elements to writing a strong clinical trial - Protocol Builder

Key elements to writing a strong clinical trial - Protocol Builder

Clinical Trials Protocol Template - Office of Science Policy - NIH

Protocol guidance and template for use in a Clinical Trial of an

Clinical Trials Protocol Template - Office of Science Policy - NIH
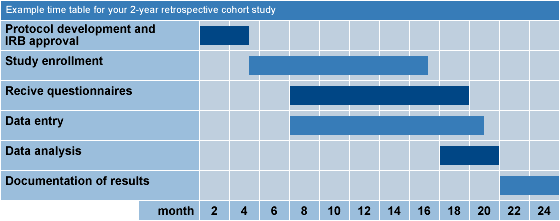
FDA and NIH Release a Draft Clinical Trial Protocol Template for

Effectively Writing Clinical Trial Protocols - Barnett Educational Services

Effectively Writing Clinical Trial Protocols - Barnett Educational Services

Key elements to writing a strong clinical trial - Protocol Builder

Protocol guidance and template for use in a Clinical Trial of an

Clinical Trials Protocol Template - Office of Science Policy - NIH
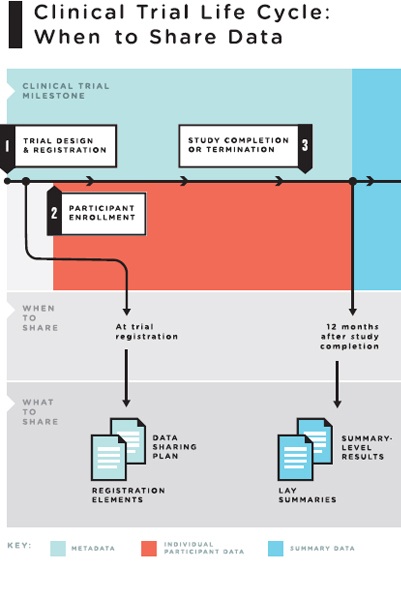
Effectively Writing Clinical Trial Protocols - Barnett Educational Services

Writing a Protocol | CHOP Institutional Review Board
